
In order to initiate fission, a high-energy neutron is directed towards a nucleus, such as ^ Fission bomb assembly methods: Two methods have been applied to induce the nuclear chain reaction that produces the explosion of an atomic bomb. These nuclei are called unstable, and this instability can result in radiation and fission. In these nuclei, it’s possible for particles and energy to be ejected from the nucleus. As the nucleus gets bigger, the electromagnetic force becomes greater than the strong nuclear force. In atoms with small nuclei, the strong nuclear force overpowers the electromagnetic force. The strong nuclear force acts to hold all the protons and neutrons close together, while the electromagnetic force acts to push protons further apart. These two forces produce opposite effects in the nucleus. The electromagnetic force causes the repulsion between like-charged protons. This force binds protons and neutrons together inside the nucleus, and it is most powerful when the nucleus is small and the nucleons are close together. The strong nuclear force is the force between two or more nucleons. Within the nucleus, there are different forces that act between the particles. The resulting elements (shown here as Kr-92 and Ba-141) do not contain as many nucleons as U-236, with the remaining three neutrons being released as high-energy particles, able to bombard another U-235 atom and maintain a chain reaction. If U-235 is bombarded with a neutron (light blue small circe), the resulting U-236 produced is unstable and undergoes fission. There are also ways to modulate the chain reaction by soaking up the neutrons. The fission process also releases extra neutrons, which can then split additional atoms, resulting in a chain reaction that releases a lot of energy. Nuclear fission: In nuclear fission, an unstable atom splits into two or more smaller pieces that are more stable, and releases energy in the process. The smallest of these fragments in ternary processes ranges from the size of a proton to the size of an argon nucleus. Occasionally, about 2 to 4 times per 1000 events, three positively charged fragments are produced, which indicates a ternary fission. Most fissions are binary fissions that produce two charged fragments. The nuclei produced are most often of comparable but slightly different sizes, typically with a mass ratio of products of about 3:2 for common fissile isotopes. The fission of heavy elements is an exothermic reaction, and huge amounts of energy are released in the process. Nuclear fission is a process by which the nucleus of an atom is split into two or more smaller nuclei, known as fission products. nuclear fission: Radioactive decay process in which the nucleus of an atom splits into lighter nuclei.nucleon: One of the subatomic particles of the atomic nucleus, i.e.fissile: Capable of undergoing nuclear fission.When neutrons are released during the fission process, they can initiate a chain reaction of continuous fission which sustains itself. 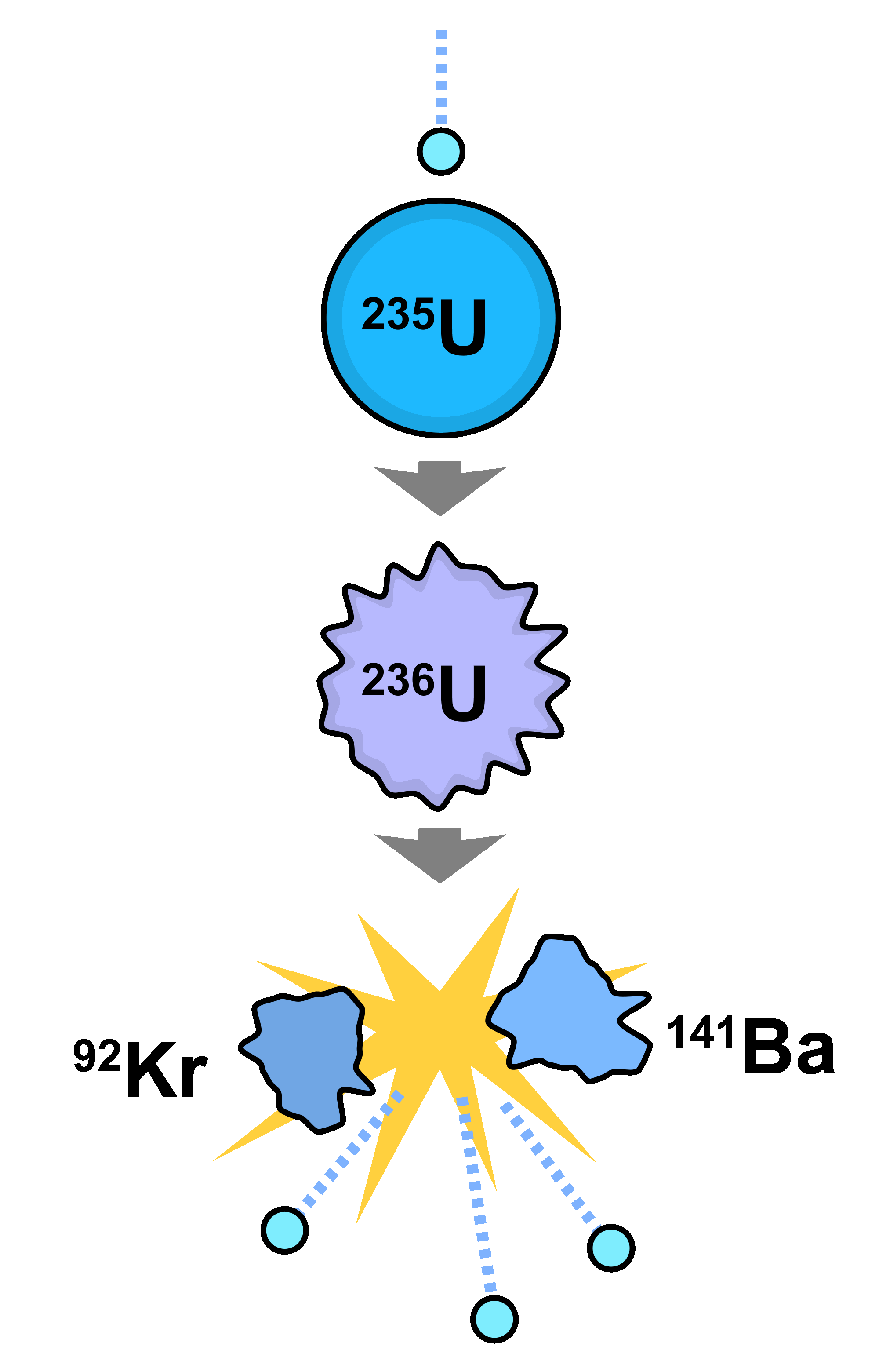
In order to initiate most fission reactions, an atom is bombarded by a neutron to produce an unstable isotope, which undergoes fission.Nuclear fission occurs with heavier elements, where the electromagnetic force pushing the nucleus apart dominates the strong nuclear force holding it together.The fission of heavy elements is an exothermic reaction, and huge amounts of energy are released in the process.Nuclear fission is a process where the nucleus of an atom is split into two or more smaller nuclei, known as fission products.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
